Moroccan Midwife Toad
Alytes maurus Pasteur and Bons, 1962
By Arlo Hinckley and Alberto Sánchez-Vialas
Updated: 30/03/2020
Taxonomy: Anura | Alytidae | Alytes | Alytes maurus

 Alytes maurus
Alytes maurus
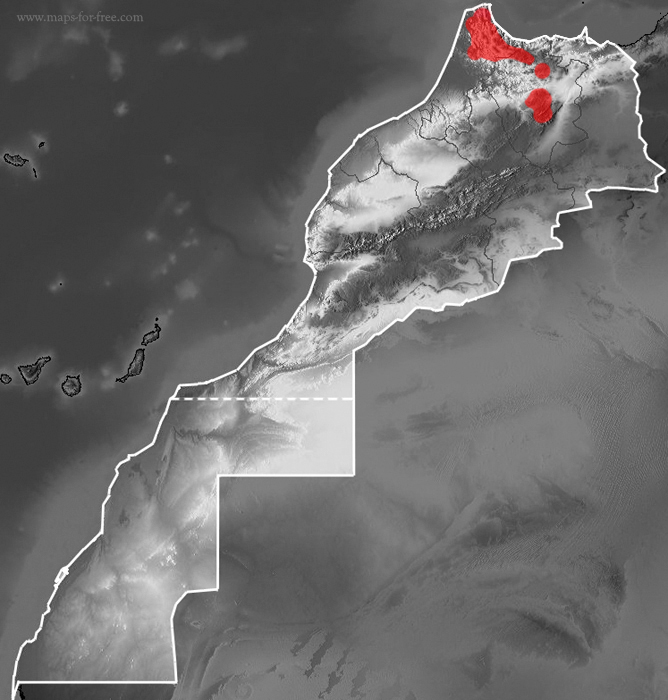
Distribution map of
Alytes maurus
in Morocco.
Phylogenetic frame
The genus Alytes includes three subgenera and six species native to the Western Palearctic: Alytes (Alytes) obstetricans (Laurenti, 1768), Alytes (Alytes) almogavarii Arntzen & García-París, 1995, Alytes (Ammoryctis) cisternasii Boscá,1879, Alytes (Baleaphryne) dickhilleni Arntzen & García-París, 1995, Alytes (Baleaphryne) muletensis (Sanchíz & Adrover, 1979) and Alytes (Baleaphryne) maurus Pasteur & Bons, 1962. The phylogenetic relationships among species of the subgenus Baleaphryne have recently been resolved (Maia-Carvalho et al. 2014). Alytes maurus represents the sister taxon of the Betic midwife toad Alytes dickhilleni, with the Majorcan endemism Alytes muletensis as a sister species of both (A. dickhilleni + A. maurus). The split between A. maurus and A. dickhilleni seems to be related to the reopening of the Strait of Gibraltar during the end of Miocene (Martínez-Solano et al., 2004).
At a population level, Alytes maurus shows no strong genetic structure (based on mitochondrial DNA) possibly due to recent gene flow among populations. Their populations would have been connected during the last glacial maximum as a result of an increased climatic suitability for the species (de Pous et al. 2013).
Description
Small and stocky frog with brownish, greenish or whitish dorsal colouration over which well defined black, green or brown spots might be present. Tiny orange dots in the body sides, parotoid glands and limbs might also be present. White ventral skin, sometimes with spots of grey on the throat and chest (Schleich et al 1996; Donaire-Barroso & Bogaerts, 2003; Escoriza & Ben Hassine, 2019).
Head large, broader than long with a rounded snout. Prominent eyes showing vertical pupil. Distinct tympanum which is smaller than the eye. Parotoid glands are very small and narrow, not always clearly visible. Completely round tongue. Short and stout limbs. Hands with short and unwebbed digits, with three metacarpal tubercles. Rudimentary webbed hind-feet with a small inner metatarsal tubercle present. The skin is granulated with small tubercles (Escoriza & Ben Hassine, 2019).



Snout-vent length (SVL) recorded from specimens of Bab Taza (Rif Mountains) were 34.1-45-2 mm for males and 36.3 and 47.4 mm for females (Escoriza & Ben Hassine, 2019). The average snout-vent length (SVL) of seven males from Jebel Tazekka was 40.2 mm (range: 38-43 mm), whereas six of these males average weight was 6.91 g (range: 6-8 g) (Márquez et al. 2011). SVL recorded for four females from Chefchaouen was 44.5 mm (Donaire-Barroso & Bogaerts, 2003). Metamorphic specimens present a SVL around 25 mm (Donaire-Barroso & Bogaerts, 2003). Sexual dimorphism is poorly developed; Escoriza & Ben Hassine (2019) noted that males have more robust forelimbs than the females.
Diagnosis: Not different in general morphology from the European species Alytes obstetricans but Salvador (1996) remarks that Alytes maurus is smaller, more slender, and with longer and thinner digits. The tadpoles have some distinctive features (Pasteur & Bons, 1962); Escoriza and Ben Hassine (2019) provide a detailed larval description and illustrations of several larval stages, including the oral disc, highlighting the labial tooth row formula.
Ecology and habits
The Moroccan midwife toad is terrestrial and nocturnal, it lives underground in rock crevices, caverns or under stones in mountain karst, mainly close to small streams.
Males call at dusk and during night, even when carrying eggs. The call is like a short whistle. Call parameters are within the range of variation of those of Alytes obstetricans, but are shorter in duration than those of Alytes cisternasii and Alytes dickhilleni (Márquez et al., 2011).
The breeding season varies among populations: calls have been heard at the beginning of February in the Tingitana Peninsula (authors pers. obs.) and from February and April in the western Rif Mountains (Donaire-Barroso & Bogaerts, 2003). Libis (1985) reported the calls in June and Bons (1967) between April and June.
It is the only North African anuran with a highly developed parental care: Alytes species have a courtship that involves male emitting advertisement calls to attract females and vocalizing females approaching the calling male. This is followed by an elaborate amplexus that occurs on land where males twine the strings with fertilized eggs around their ankles and carry them for about 3 weeks. Once the tadpoles internal gills are developed, the male proceeds into water, releasing the eggs which will hatch into tadpoles. Clutch size is around 25-30 eggs (Escoriza & Ben Hassine, 2019), with records such as 35 on a male found in Jbel Tazekka (1283 m a.s.l) and 60-70 in one from Jbel Ajnane (500 m a.s.l) (Donaire-Barroso & Bogaerts 2003; Márquez et al. 2011).



Larvae-bearing populations of S. a. tingitana and S. a. splendens are often found in sympatry with Alytes maurus. Other syntopic species are Bufo spinosus, Pelophylax perezi, Sclerophrys mauritanica, Hyla meridionalis and Discoglossus scovazzi (Beukema et al. 2013; authors pers. obs.).
Distribution, habitat and abundance
Alytes maurus is found in North Morocco in the Rif Mountains (where is located the type locality=Talassemtane) and in the Middle Atlas Mountains. In the Middle Atlas it seems to be limited to the Tazekka and Bouiblane Massifs. The altitudinal distribution ranges from 200 up to 2142 m a.s.l., with most of the observations localized at intermediate elevation (Donaire-Barroso & Bogaerts, 2003).
Although A. maurus was cited in Beni Snassen massif, their presence in this region has not been confirmed during different surveys performed in this area (Dinis et al., 2019; Escoriza & Ben Hassine, 2019). In north-western Algeria, the presence of this species is considered suitable according to the models of potential distributions (de Pous et al. 2013); however, no direct evidence of this species has been published for Algeria. Mateo et al. (2013) listed Alytes as part of the amphibian fauna of Algeria based on two adult conserved specimens which were incorrectly identified and labelled at the collection of the Asociación de Amigos de Doñana and some tadpoles sighted in Hafir Forest with sign of Chytridiomycosis. Most authors did not follow this record and excluded it from the amphibian fauna of Algeria (Escoriza & Ben Hassine, 2019) until more evidence was provided. In fact, recent surveys in Tlemcen by experienced local herpetologists have not detected this species (Khaled Merabet personal communication).
Alytes maurus inhabits grasslands, forest, stone walls close to agricultural fields, and rocky valleys, usually near water sources (Beukema et al., 2013; Escoriza & Ben Hassine, 2019; Mabrouki et al., 2019). In the Rif, it occupies areas dominated by Quercus suber, Quercus ilex, Quercus pyrenaica, Juniperus oxycedrus, Chamaerops humilis, Nerium oleander, Crataegus monogyna and Olea europaea. Populations in the Middle Atlas are encountered in rocky areas with bushy thorny vegetation, and Pinus, Quercus, and Cedrus atlantica forests (Donaire-Barroso et al., 2006; Beukema et al., 2013).



It is listed as Near Threatened (Donaire-Barroso et al., 2009). However, its limited distribution makes it highly vulnerable and might suppose a shift towards a more endangered status in the following years. Different studies agree on a negative trend for this species driven by the following threats: deforestation, invasive species, contamination of water, climate change and emergent diseases. In the Rif Mountains, peri-urban populations might suffer important declines due to a combination of water pollution, overgrazing, and introduction of Gambusia holbrooki (Mateo et al., 2003; Donaire-Barroso et al., 2009) In fact, Márquez et al. (2011), only recorded calling activity in one over seven known populations of Alytes during a five-year study. Size reduction of breeding sites due to human water extraction and a sharp decrease in larvae density point towards potential local extinctions in the nearby future. In the Tingitana Peninsula, some population’s reproductive habitats are highly polluted by littering (authors pers obs). Middle Atlas populations outside of protected areas are also subjected to similar threats such as cattle grazing, deforestation, canalization of mountain water, and soil erosion (Donaire-Barroso et al., 2006). De Pous et al., (2013) showed that only about 7% of the distribution of A. maurus falls inside protected areas although many remaining populations are in areas so rugged that alteration is not imminent (Donaire-Barroso and Bogaerts, 2003). Finally, the devastating effects of the chytrid fungus (El Mouden et al., 2011) and climate change are two major drivers threatening populations within and outside protected areas. Rodriguez-Rodriguez et al. (2020) considered A. maurus a species with underestimated conservation priorities given that different climate change scenario models predicted a decline in suitable habitat.To sum up, the vulnerability of this species is possibly underestimated (Pleguezuelos et al., 2010; Mabrouki et al., 2019) and an increase in monitoring efforts should be performed to assess trends and conservation priorities (Slimani et al., 2010).
Bibliography
- Arntzen, J. W., García-París, M. 1995. Morphological and allozyme studies of midwife toads (genus Alytes) including the description of two new taxa from Spain. Contrib. Zool., 65, 5-34.
- Beukema, W., De, P. P., Donaire-Barroso, D., Bogaerts, S., Garcia-Porta, J., Escoriza, D., Carranza, S. 2013. Review of the systematics, distribution, biogeography and natural history of Moroccan amphibians. Zootaxa, 3661, 1-60.
- Bons, J. 1972. Herpetologie Marocaine 1. Liste commentee des amphibiens et reptiles du Maroc. Bulletin de Société des Sciences Naturelles et Physiques du Maroc, 52, 107–126.
- Bons, J., Geniez, PH. 1996. Amphibiens et Reptiles du Maroc (Sahara Occidental compris). Atlas biogéographique. AHE, Barcelona. 320 pp.
- de Pous, P., Metallinou, M., Donaire-Barroso, D., Carranza, S., Sanuy, D. 2013. Integrating mtDNA analyses and ecological niche modelling to infer the evolutionary history of Alytes maurus (Amphibia; Alytidae) from Morocco. The Herpetological Journal, 23(3), 153-160.
- Dinis, M., Merabet, K., Martínez-Freiría, F., Steinfartz, S., Vences, M., Burgon, J.D., Elmer, K.R., Donaire, D., Hinckley, A., Fahd, S. and Joger, U., 2019. Allopatric diversification and evolutionary melting pot in a North African Palearctic relict: the biogeographic history of Salamandra algira. Molecular phylogenetics and evolution, 130, 81-91.
- Donaire-Barroso, D., and S. Bogaerts. 2003. Datos sobre taxonomía, ecología y biología de Alytes maurus (Pasteur & Bons, 1962) (Anura, Discoglossidae). Butlletí de la Societat Catalana d’Herpetologia. Barcelona 16:25–40.
- Donaire-Barroso, D., El Mouden, E. H., Slimani, T., de la Vega, J. P. 2006. On the meridional distribution of Alytes maurus Pasteur and Bons, 1962 (Amphibia, Discoglossidae). Herpetological bulletin, 96, 12.
- Donaire-Barroso, D., Salvador, A., Slimani, T., El Mouden, E.H, Martínez-Solano, I. 2009. Alytes maurus. In IUCN 2012. IUCN Red List of Threatened Species. Version 2012.2. Downloaded on 30 October 2012.
- El Mouden, H., Slimani, T., Donaire, D., Fernandez-Beaskoetxea, S., Fisher, M., Bosch, J. 2011. First record of the chitrid fungus Batrachochytrium dendrobatidis in North Africa. Herpetological Review 42: 71.
- Escoriza D, Ben Hassine J. 2019. Amphibians of North Africa. Academic Press, London, 350 pp 10.1016/B978-0-12-815476-2.00002-X
- Harris, D.J., Carretero, M.A., Brito, J.C., Kaliontzopoulou, A., Pinho, C., Perera, A., Vasconcelos, R., Barata, M., Barbosa, D., Carvalho, S., Fonseca, M.M., Pérez-Lanuza, G., Rato, C. 2008. Data on the distribution of the terrestrial herpetofauna of Morocco: Records from 2001-2006. Herpetological Bulletin, 103, 19–28.
- Mateo, J., Geniez,P., Pether, J. 2013. Diversity and conservation of Algerian amphibian assemblages. Basic and applied Herpetology, 27, 51-83.
- Maia-Carvalho, B., Gonçalves, H., Ferrand, N., Martínez-Solano, I. 2014. Multilocus assessment of phylogenetic relationships in Alytes (Anura, Alytidae). Molecular phylogenetics and evolution, 79, 270-278.
- Libis, B. 1985. Nouvelle donnée sur la répartition au Maroc du crapaud accoucheur Alytes maurus (Amphibia; Discoglossidae). Bull. Soc. Herp. Fr. 33: 52-53.
- Loukkas, A. 2006. Atlas des Parcs Nationaux Algériens. République Algerienne Démocratique et Populaire, Ministere de l’Agriculture et du Developpement Rural, Direction Générale des Forêt et Parc National de Théniet El Had, Alger, Algeria. Available at http://www.fichier-pdf.fr/2010/12/19/atlas-des-parcs-nationaux/. Retrieved on 07/19/2009.
- Mabrouki, Y., Taybi, A. F., Skalli, A., Sánchez-Vialas, A. 2019. Amphibians of the Oriental Region and the Moulouya River Basin of Morocco: distribution and conservation notes. Basic and Applied Herpetology, 33, 19-32.
- Márquez, R., Francisco Beltrán, J., Slimani, T., Radi, M., Llusia, D., El Mouden, H. 2011. Description of the advertisement call of the Moroccan midwife toad (Alytes maurus Pasteur & Bons, 1962). Alytes, 27(4), 142-150.
- Pleguezuelos, J. M., Brito, J. C., Fahd, S., Feriche, M. F., Mateo, J. A., Moreno-Rueda, G., Reques, R., Santos, X. 2010. Setting conservation priorities for the Moroccan herpetofauna: the utility of regional red listing. Oryx, 44 (4): 501-508.
- Rodríguez-Rodríguez, E. J., Beltrán, J. F., El Mouden, E. H., Slimani, T., Márquez, R., Donaire-Barroso, D. 2020. Climate change challenges IUCN conservation priorities: a test with western Mediterranean amphibians. SN Applied Sciences, 2(2), 1-11.
- Salvador, A., 1996. Amphibians of northwest Africa. Smithson. herp. Inf. Serv., 109: 1-43.
- Slimani, T., Lagarde, F., El Mouden, E. H., Bonnet, X., Lourdais, O., Benkaddour, K., Márquez, R., Beltran, J. F., 2010. The Moroccan Herpetology. Basic research to the conservation of species. Atti. VIII Congresso Nazionale Societas Herpetologica Italica: 159-168.
To cite this page:
Hinckley A and Sánchez Vialas A. 2020. Alytes maurus Pasteur and Bons, 1962. In: Martínez, G., León, R., Jiménez-Robles, O., González De la Vega, J. P., Gabari, V., Rebollo, B., Sánchez-Tójar, A., Fernández-Cardenete, J. R., Gállego, J. (Eds.). Moroccoherps. Amphibians and Reptiles of Morocco.
Available from www.moroccoherps.com/en/ficha/alytes_maurus/. Version 9/09/2015.
To cite www.morocoherps.com en as a whole:
Martínez, G., León, R., Jiménez-Robles, O., González De la Vega, J.P., Gabari, V., Rebollo, B., Sánchez-Tójar, A., Fernández-Cardenete, J.R., Gállego, J. (Eds.). Moroccoherps. Amphibians and Reptiles of Morocco.
Available from www.moroccoherps.com.

